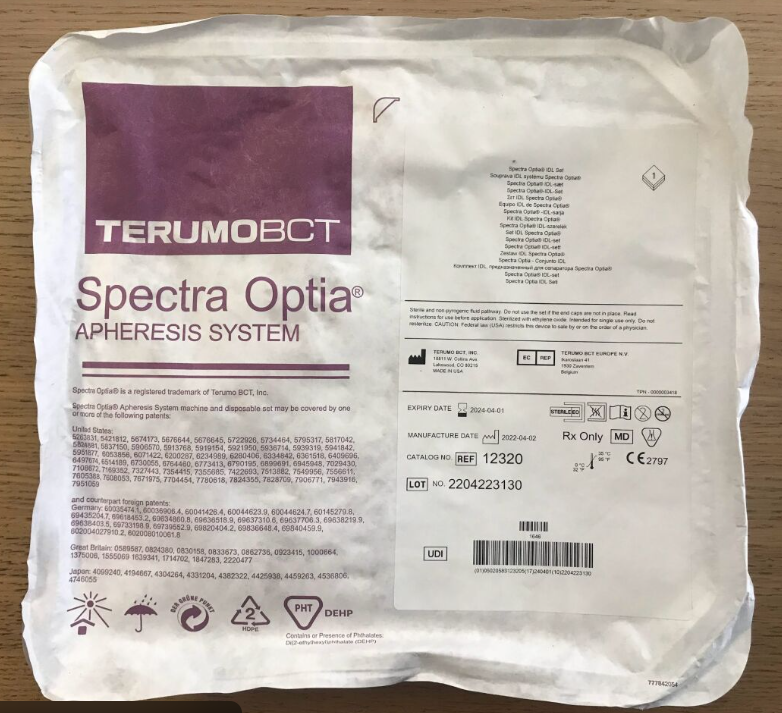
Terumo BCT
SPECTRA OPTIA IDL SET 12320
Technical characteristics
Manufacturer:
Terumo BCT, Inc.
Product Name:
One-time set of SPECTRA OPTIA IDL mains
Catalog number:
12320
Compatible Hardware:
The automated apheresis system of the Spectra Optia Apheresis System
1. Purpose
The disposable IDL kit is designed to perform apheresis and therapeutic extracorporeal procedures using the Spectra Optia system, including collecting, processing, separating and returning blood components from a patient or donor.
2. Scope of application
- blood services;
- transfusion departments;
- Departments of Hematology and Oncohematology;
- intensive care units;
- medical and preventive institutions using the Spectra Optia system.
3. Type of procedures
- therapeutic plasmapheresis;
- therapeutic erythrocytopheresis;
- therapeutic leukapheresis;
- plasma Exchange (TPE);
- other therapeutic procedures within the framework of IDL protocols.
4. The composition of the kit
The disposable kit includes:
- highway system for extracorporeal circuit;
- venous lines (arterial and venous);
- pump segments compatible with Spectra Optia roller pumps;
- containers (bags) for collecting and removing components;
- connecting elements, clamps and valves;
- sections of highways intended for thermal sealing.
5. Needles and venous access
- Type: Disposable venous needles
- Caliber: 16G
- Material: Medical grade stainless steel
- Connection: integrated into the backbone system
- Purpose: provision of venous access
6. The backbone system
- Tube material: Medical grade PVC, biocompatible
- Inner diameter of tubes: ≈ 3.0–4.0 mm (depending on segment)
- Pumping segments: provided by
- Clamps: built-in, mechanical
- Connections: sealed, factory assembled
7. Containers (bags)
The composition and volumes depend on the selected protocol of the procedure
- bags for collecting removable components;
- bags for temporary storage of plasma or other fractions;
- Nominal volumes: 300 300-1000 ml
- Material: medical polymer compatible with blood components
- Type: Disposable
8. Sterility and safety
- a sterile, non-pyrogenic product;
- it is intended exclusively for one-time use;
- reuse and repeated sterilization are prohibited.;
- provides a closed sterile extracorporeal system.
9. Operational characteristics
- Type of blood contact: extracorporeal
- Operating mode: continuous
- Usage: by qualified medical personnel who have been trained to work with the Spectra Optia system.
10. Storage conditions
- Storage temperature: from +2 °C to +30 °C;
- store in a dry place protected from light;
- use only in complete original packaging and within the expiration date.
11. Classification
- medical device for extracorporeal blood treatment;
- disposable consumables;
- a component of the Spectra Optia automated apheresis system.